PURPOSE - Built for Regulated Industries.
In-Q Center delivers expert-validated, citation-backed answers from U.S. regulatory frameworks and global standards, built for FDA- and USDA-regulated industries and customizable to your organization’s requirements.
Regulatory Intelligence
Audit-Ready Answers
Policy Integration
Multi-Lingual Coverage
Expert Validated
Instant Results
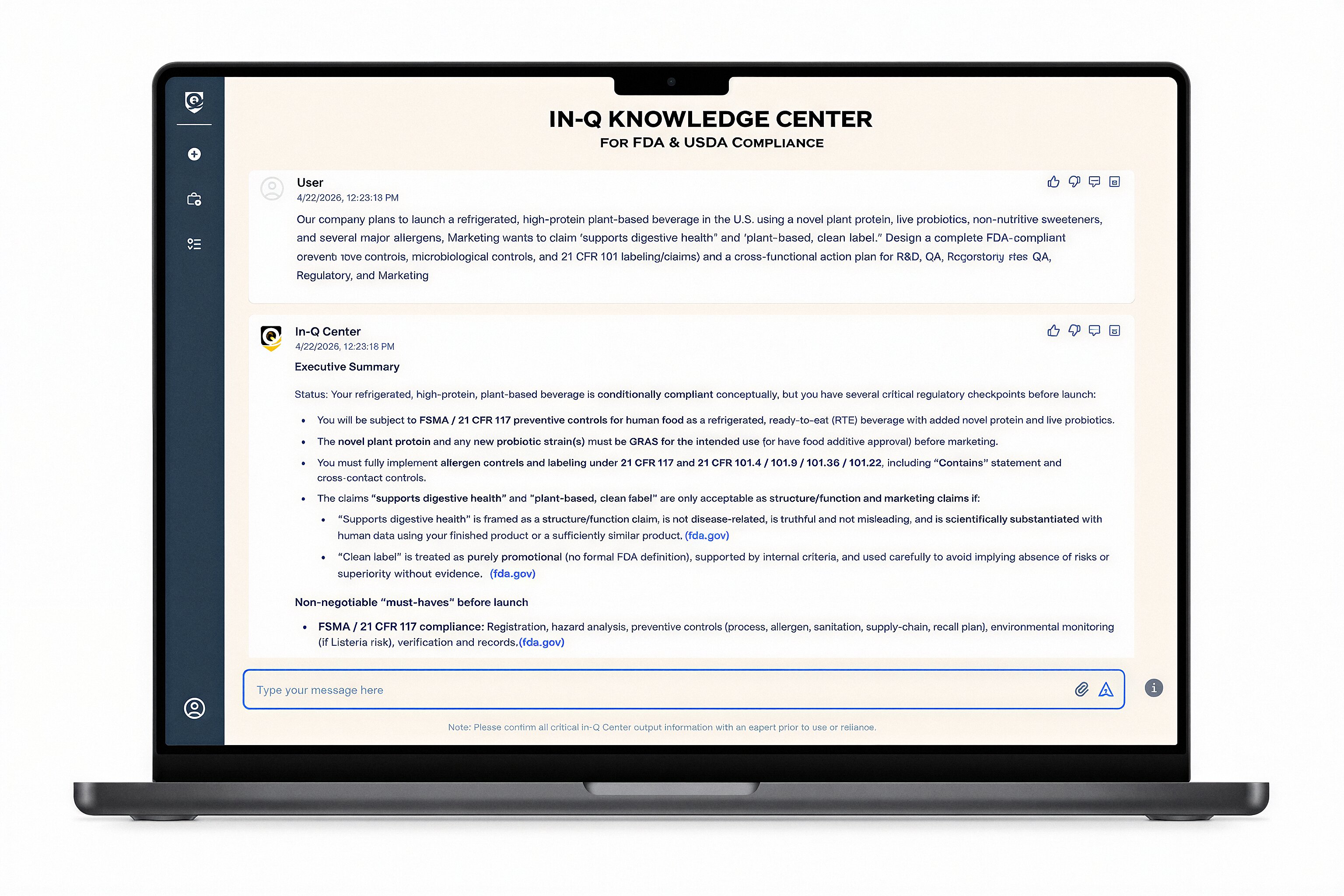
One Decision Can Change How Your Business Operates
Move from slow, manual workflows to clear answers and faster decisions across your team.
Teams Without In-Q Center
Weeks disappear into document review. Consultants drain budgets. Risk stays high.
Spend hours searching through regulations and documents
Rely on internal knowledge or external consultants
Review the same information multiple times
Second-guess decisions before moving forward
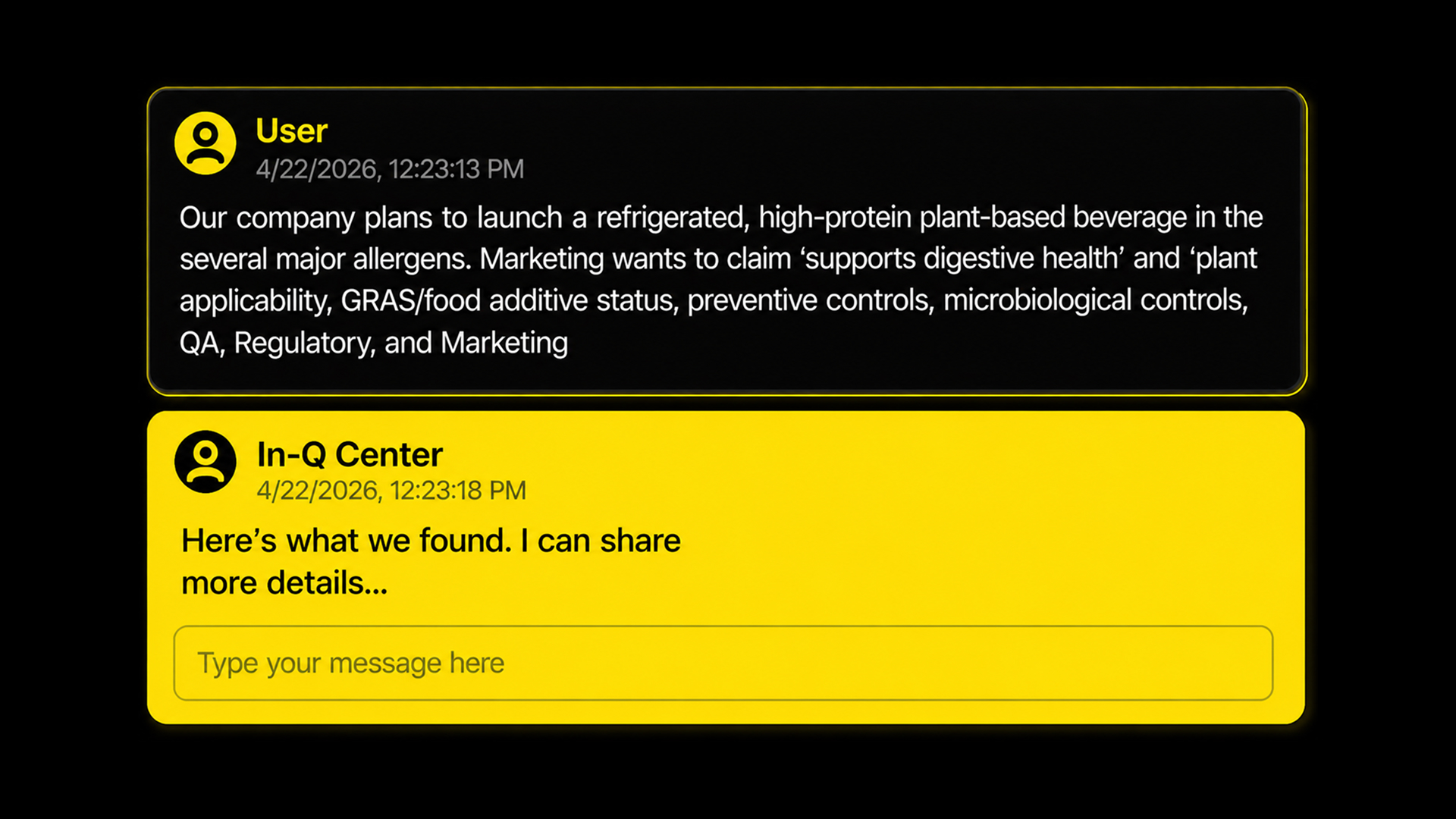
Teams With In-Q Center
Weeks disappear into document review. Consultants drain budgets. Risk stays high.
Get clear, citation-backed answers in seconds
Use experts for validation of information only
Instantly find what matters in complex documents
Move forward with compliant actions and consistency
How In-Q Center Transforms Your Process
In-Q Center reduces the manual burden of regulatory research, interpretation, and document review—so your teams can move faster, make better decisions, and stay aligned with compliance requirement
Personalized Responses
Tailor outputs based on your role, regulatory scope, and use case.

Analyze Regulatory Inputs
Analyze questions and documents using AI models trained on real regulatory frameworks.
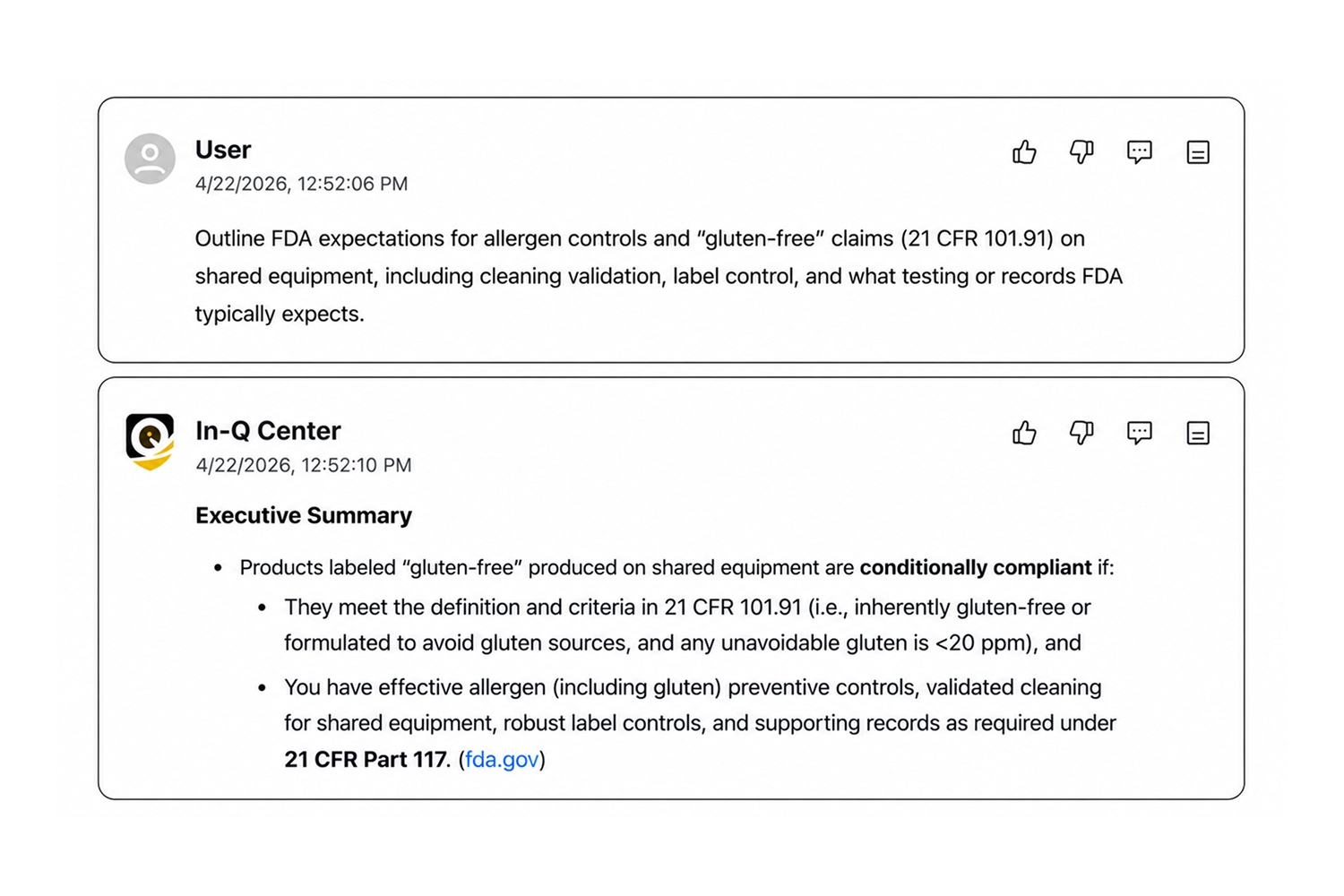
Citation-Backed Answers
Every answer includes precise citations for verification and auditability.

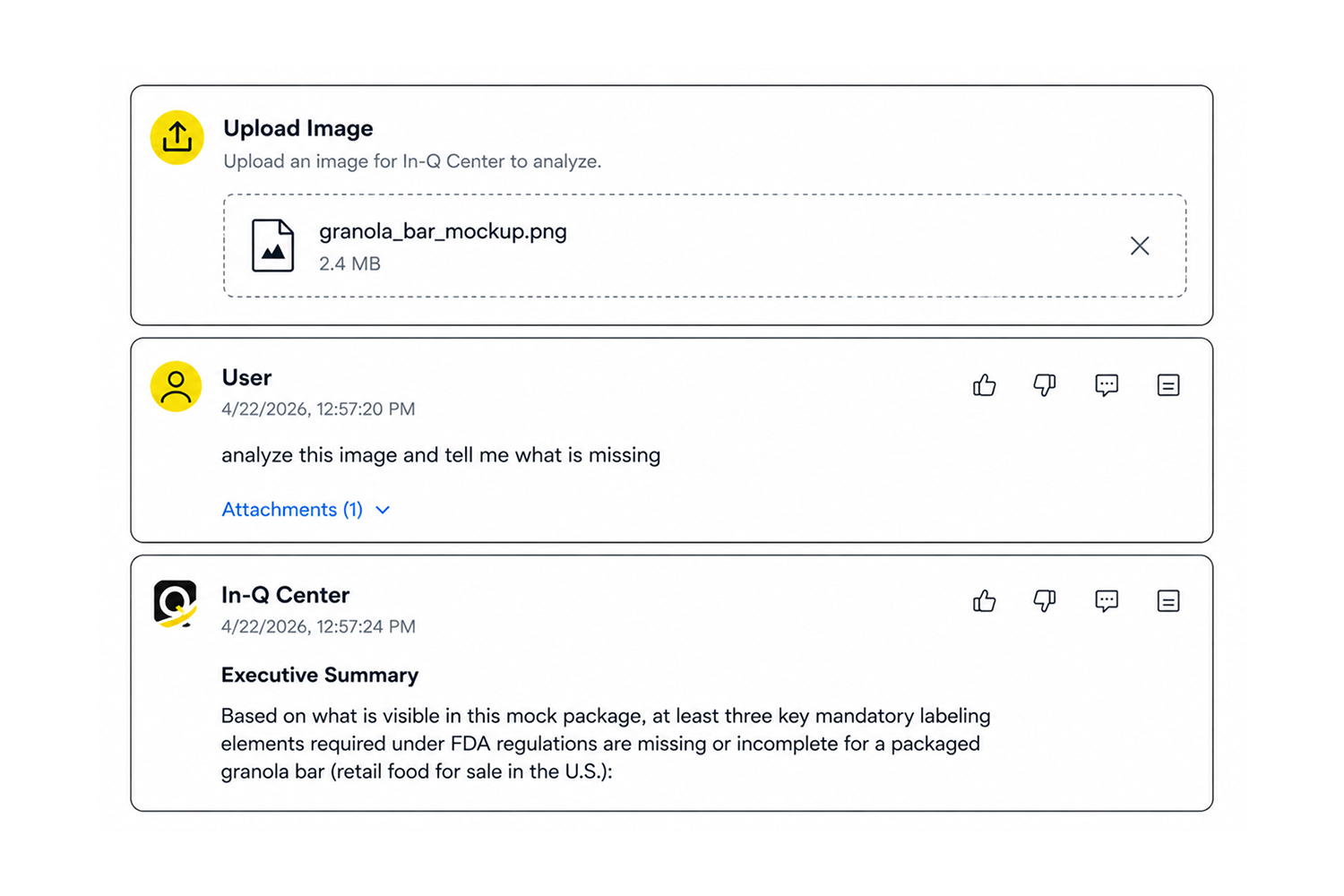
Validate Labels and Product Data
Upload product labels or specs to check compliance against regulatory requirements.
Built For Secure and Compliant Regulatory Decisions
Intended for use in an enterprise setting.
Data Encryption
End-to-end encryption for data in transit and at rest using industry-standard protocols.
Access Control
Multi-factor authentication to ensure only authorized access.
Audit & Traceability
Complete audit trails, activity logging, and version history for full transparency and accountability.
Data Privacy
Data isolation, secure processing, and privacy-by-design architecture to protect sensitive information.
Enterprise Compliance
Built for global regulatory and enterprise security standards.
From Manual Work to Measurable Results
Built to transform regulatory complexity into practical guidance your teams can quickly act on.
FAQs
Common questions about In-Q Center.
In-Q Center is an AI-powered regulatory intelligence platform designed to help teams quickly and confidently access, interpret, and work with U.S. regulatory requirements and global standards in a structured, auditable way. The tool is designed to provide action-oriented recommendations for cross-functional teams, helping ensure regulatory and compliance standards are consistently and accurately applied across your supply chain and operations.
n-Q Center is built for FDA- and USDA-regulated industries, including food and beverage, ingredients, flavors and fragrances, dietary supplements, packaging, pet food, and other regulated manufacturing sectors.
Yes. In-Q Center provides citation-backed answers linked to the original regulatory sources and standards to support traceability, verification, and audit readiness.
Yes. Enterprise configurations can include your internal standards, SOPs, policies, customer requirements, approved workflows, and other organization-specific content.
Yes. While the platform’s validated foundation is centered on U.S. regulatory requirements, In-Q Center also provides access to global standards and international regulatory references. The platform allows easy comparisons between regional and country regulations, as well as your internal standards.
In-Q Center reduces manual searching, document creation, review, comparison, and repetitive regulatory interpretation tasks, helping teams respond faster, improve consistency, and make more informed compliance decisions across operations.
No. Customer data is never used to train our AI models. In-Q Center is designed with enterprise security, controlled access, and data privacy in mind.
Yes. Regulatory, Quality, Food Safety, R&D, Procurement, Manufacturing, and other cross-functional teams can all leverage the platform based on their workflows and access permissions.
In-Q Center is designed with enterprise-grade security controls, including encrypted data handling, role-based access, audit traceability, and controlled environments to help protect sensitive regulatory and operational information. Customer data is not used to train public AI models.
Still have questions?
Talk to our team to see how Innova-Q products fit into your workflow.
Ready to move faster?
Let Innova-Q handle the complexity. Your team focuses on what matters.

